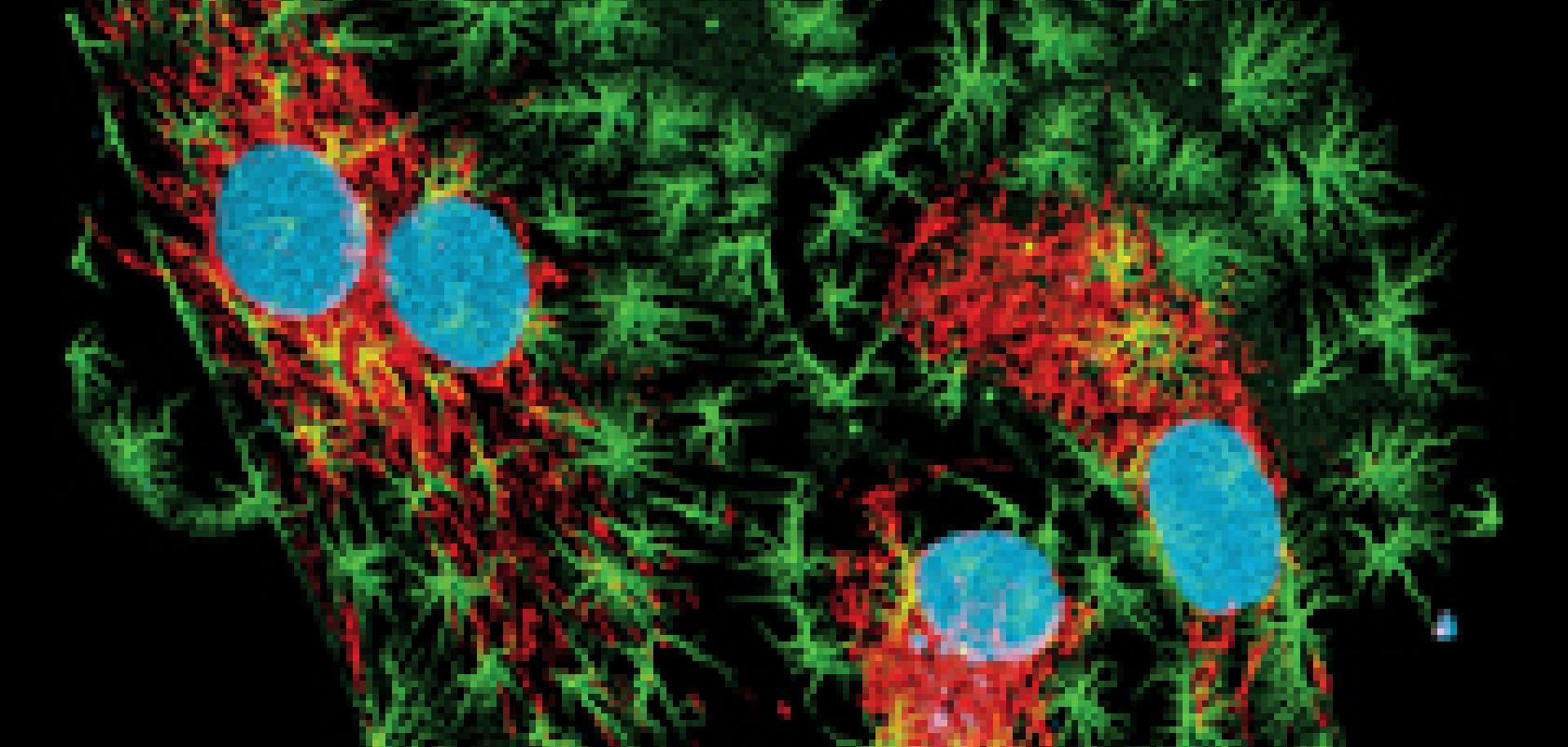
In recent years, super-resolution fluorescence microscopy has evolved at a fast pace; scientists can now image at a nanometre scale to observe molecular interactions and dynamic processes.
One limitation of fluorescence microscopy, however, is that the red fluorescent probes used for labelling tissue specimens are not very bright or efficient. To solve this, researchers are developing a new group of near-infared fluorescent proteins for enabling greater tissue depths with improved sensitivity.
Meanwhile, in separate research, scientists are advancing label-free live imaging techniques that, for certain applications, eliminate the drawbacks associated with using fluorophores, such as phototoxicity.
The availability of bright fluorescent markers that can highlight molecules and structures specifically and efficiently is central to successful fluorescence microscopy. For live imaging, the green fluorescent protein (GFP) family of markers have become the gold standard, because of their ability to be genetically encoded and expressed in cells, resulting in a fusion construct with the protein of interest.
This provides a powerful and versatile technique for labelling live samples. Since its discovery in the 1960s, scientists have worked on optimising marker proteins of the GFP family, and the colour palette has been extended from the blue to the far-red side of the spectrum. It’s safe to say that the green fluorescent protein and its colour-shifted genetic derivatives have revolutionised live cell imaging.
Far-red emitting fluorescent proteins (FPs) are advantageous as markers for live specimens for multiple reasons. Not only is there less absorption, scattering and autofluorescence in the red part of the spectrum, but red light is less phototoxic, and far-red FPs offer the possibility of realising an additional detection channel for multi-colour imaging.
However, despite the urgent need for far-red FPs, researchers have not yet succeeded in engineering markers that can compete with the performance of GFP. Existing far-red FPs do not go far enough into the infrared to emit a bright enough signal; currently, the most far red-shifted derivatives of the GFP family emit a maximum of 670nm fluorescence and excite at around 610nm. ‘Our erythrocytes contain haemoglobin, which absorbs light below 650nm,’ said Professor Vladislav Verkhusha from the Albert Einstein College of Medicine. ‘To excite and detect fluorescence efficiently in biological tissue we need fluorescence proteins with both excitation and emission spectra above 650nm.’
So, although the maximum emission value is 670nm, the excitation is still only 610nm, which isn’t ideal for imaging tissue. ‘They work partially but not perfectly,’ Verkhusha noted. ‘Fluorescence that emits below 650nm will not be visible, because it will be absorbed by haemoglobin in the tissue. There have been many research groups over the last two decades that have tried to improve the excitation and emission in the near-infrared (NIR), but this is the maximum that is achievable now.’
Professor Verkhusha and his team at the Albert Einstein College of Medicine have developed a new group of NIR fluorescent proteins based on a different family of proteins, which have excitation spectra up to 700nm and emission up to 720nm.
Derived from the phytochrome photoreceptor family found in plants, bacteria, and cyanobacteria, these proteins make it possible to image much deeper into mammalian tissue and with a higher signal to autofluorescence background ratio. ‘In our experiments, we can detect NIR fluorescent proteins at a depth of 18mm,’ said Verkhusha. ‘If you compare this depth, the signal to background ratio is 20-fold higher for NIR fluorescent proteins as compared to the most far-red shifted protein from the GFP family. This means a 20-fold better sensitivity, or 20-fold smaller number of cells we can detect.’
Whereas deep cell imaging of live cells is typically associated with two-photon excitation, the NIR fluorescent proteins can use single-photon excitation techniques because they already absorb in the NIR. Two-photon microscopy is generally chosen for its ability to achieve higher resolutions with less photobleaching than single-photon excitation techniques such as confocal microscopy. Two-photon lasers excite by using near simultaneous absorption of two long wavelength (around 800nm) photons and, because excitation only happens near the focal plane where the laser light is most concentrated, there is little tissue damage to the surrounding areas and a higher signal to background ratio.
However, two-photon microscopy has an imaging depth limit of around several hundred micrometres, according to Verkhusha. So, in order to take advantage of the 18mm depths that these new NIR probes offer, techniques such as structured illumination, single-sheet illumination, adaptive optics microscopy, and photoacoustic imaging can be used. ‘These new imaging techniques will allow users to have high resolution in deep tissue using one-photon NIR excitation. They use lower power than two-photon microscopy, so there is less damage to the tissue. Because of this low level of damage, live tissue imaging is more feasible in one-photon regimes with NIR probes,’ Verkhusha noted.
‘As a result, I think these imaging techniques will become more popular as biologists adopt these new probes, because two-photon microscopy has existed for about 20 years and, during this time, I have not seen much development,’ Verkhusha continued. ‘These days, wavefront engineering imaging techniques are becoming more and more popular. NIR fluorescent proteins, which we develop, fit well with them.’
Label-free imaging
As well as engineering dyes that are more compatible with live tissue, scientists are improving the methods for imaging live cells without the use of labels.
Whereas fluorescence microscopy is often the method of choice for observing structures or dynamic processes in biological samples, in some cases, labelling isn’t practical. ‘There are certain situations where you just cannot label – for example, for medical diagnostic applications where the labels could be toxic, or when it is simply too labour-intensive to label and more convenient to image in a label-free fashion,’ explained Dr Chistian Freudiger, vice president of research and development at Invenio Imaging, which develops technology for non-destructive microscopy.
‘If scientists want to image very small molecules – for example, metabolites such as glucose – the components are a lot smaller than the labels you would need to attach for imaging. If you were to label it, you would really perturb the function of these molecules that you’re trying to study,’ Freudiger said. ‘On occasions where you can’t do it, you need to start looking at label-free methods – the goal is to image in a similar way to fluorescence, but without having to add any dyes or stains.’
Coherent Raman scattering (CRS) microscopy is a dye-free method that images structures by displaying the characteristic intrinsic vibrational contrast of their molecules. The major benefit of this method is that the sample remains almost unaffected.
The technique is based on spontaneous Raman scattering, which has been used in analytical R&D applications for years but typically produces very weak signals, resulting in slow acquisition rates in imaging applications. ‘The big issue with this method is the limited sensitivity – you need to wait a long time before you can get a reliable measurement,’ said Freudiger.
CRS provides amplification of the weak spontaneous Raman signal to enable high-speed, label-free chemical imaging. ‘With coherent Raman scattering we take the spontaneous signal and we amplify it by many orders of magnitude – i.e. by a factor of 10,000. So, suddenly, we can do this label-free measurement at high enough speeds to use it in microscopy.’
In general, coherent Raman scattering refers to two independent, but related, techniques: coherent anti-stokes Raman scattering (CARS) and stimulated Raman scattering (SRS), which both have the speed advantage over spontaneous Raman scattering.
As a research assistant and then postdoctoral fellow working with Professor Xiaoling Sunney Xie (developer of CARS) at Harvard University, Freudiger co-invented SRS microscopy with colleagues at the Xie lab. Invenio Imaging was founded in 2012 based on Professor Xie’s research, and over the last two years the company has produced a research-based system that can be operated in a similar way to a confocal or two-photon fluorescence microscope, but obtains an SRS contrast.
The mechanism of the contrast, Freudiger explained, is based on the vibrational frequencies of the molecules: ‘If you have two atoms, they are connected with a spring, and they’ll have a resonance frequency determined by how heavy the atoms are and how stiff the spring is in between,’ he said. ‘By looking at where the vibration is coming from, you know what the molecule is.’
What is specific to coherent Raman scattering is that, instead of using a single wavelength laser as with spontaneous Raman scattering, two-wavelength lasers are used. ‘If you fire these two wavelengths into the sample, the intensity that results is the beat frequency – the difference in frequency of the two laser wavelengths,’ explained Freudiger. ‘If you now match this beat frequency to the vibrational frequency then you can “shake” the molecules – so, you very actively drive the vibrations. What you measure in CARS is the emission that then originates from these vibrational frequencies. What one does in SRS is look at the energy transferred to the sample that results from exciting these vibrations.’
In an area of crossover between fluorescence and label-free techniques, scientists are working on developing dyes for use with this traditionally label-free approach. Professor Wei Min from Columbia University has invented new types of Raman label, which provide the specificity advantage of labelling, but that are much smaller (a few atoms in size) so eliminate some of the drawbacks of labelling molecules. ‘They are still not as bright as typical fluorophores, but they are less disruptive to the system, because it is just very small molecules that you would add,’ Freudiger remarked. ‘So, there is this overlap region where people do use labels, together with Raman imaging, and the motivation is the same – to perturb the system that they are trying to study the least.’
Minimising tissue damage
Although label-free imaging leaves the samples largely unaffected, the technique acts more as an alternative for when labelling is not practical, as Freudiger pointed out, rather than as a complete replacement to fluorescence imaging.
Fluorophores are still a vital part of imaging live tissue and, for most fluorescence microscopy techniques, it is almost impossible to eliminate damage to the sample. ‘If you use a lamp, laser or LED there will always be some sample damage – phototoxicity – as well as photobleaching because once fluorophores are hit with light they not only emit fluorescence, but they also lose efficiency to release fluorescence and eventually photobleach,’ said Dr Kavita Aswani, senior applications scientist for life sciences, marketing at light source provider Excelitas. ‘The question is how to minimise photobleaching, because there is no way to truly avoid it.’
According to Aswani, minimising sample damage while imaging live cells ultimately comes down to limiting the sample’s exposure to light, with shutters in the case of lamps, and turning LEDs off when the sample is not being imaged. The use of pulsed lasers or pulsed LEDs may also reduce sample damage, she added. It is thought that this could be due to the ability of the cell to recover from being exposed constantly to light, thereby generating fewer reactive oxygen species. ‘So, you can give the cells time to recover before you blast them again with light, which leads to reduced phototoxicity,’ said Aswani.
For advanced microscopy applications, Excelitas has demonstrated that it’s X-Cite XLED1 high-power light source, when operated in live cell mode or pulsed mode, results in lower phototoxicity in live cells when compared to continuous light exposure. In a separate study, the company also found that cell proliferation is higher when a sample is imaged with LED than when imaged with a mercury lamp for the same exposure time, although these have only been initial findings.
But although pulsed light is typically associated with less photodamage, the extremely high speeds of multiphoton lasers can also create issues concerning sample damage.
Since two-photon excitation is a non-linear process, scientists quickly realised that shorter laser pulses at the sample resulted in higher fluorescence signal because of the higher peak power. ‘For this reason, lasers designed for multiphoton microscopy typically have pulse durations between about 50 and 150 femtoseconds,’ said Marco Arrigoni, director of marketing for Coherent’s scientific market segment. ‘However, with some biological specimens, if not properly managed, the shorter pulses also produce the undesirable effect of a higher rate of photobleaching.’
Very short pulses (i.e. 50-100fs) are more difficult to manage since their larger bandwidth results in a higher material dispersion; by the time the pulses travel through all the microscope optics and reach the sample, their duration may be longer than at the laser output. ‘Because of this, lasers like Coherent’s Chameleon Discovery and Vision incorporate a so-called pre-chirp feature that conditions the laser pulses to have the shortest duration right at the sample plane,’ Arrigoni said, adding that the laser pre-chirp feature can also be used to detune the pulses and make them purposely longer, so that the user can find the optimum pulse duration for different experiments.
Interestingly, the ideal laser parameters for minimising tissue damage in three-photon imaging, a newer technique that enables greater imaging depths in the brain, are quite different than with two-photon microscopy. Professor Chris Xu and co-workers at Cornell University recently reported that higher energies at lower repetition rates, with correspondingly lower average power, are paramount for minimising thermal sample damage, Arrigoni pointed out. ‘This newer imaging technique is leading to the adoption of lasers, like the Coherent Monaco, which produce a more energetic pulse train at 1MHz repetition rate rather than the 80MHz of more conventional mode-locked lasers,’ commented Arrigoni.
Earlier this year, UK start-up company IoLight launched a digital microscope powerful enough to observe the structure of plant and animal cells, but that can fit inside a jacket pocket.
Capable of achieving resolutions of 1µm, it unfolds to record and share 5 megapixel still images and real-time high-definition video at a magnification of x200 on an iPad.
There have been digital microscopes available on the market for several years… these use high quality mobile phone lenses and optics to produce a small, low cost microscope that is fine for low magnification applications, such as looking at insects,’ commented IoLight co-founder Andrew Monk. ‘However, these microscopes cannot see smaller subjects, like individual cells, because they are handheld and it is impossible to hold your hand still enough. They can be mounted on a robust stand, but then they are not really portable anymore.’
To make the microscope truly portable, IoLight has used these same components, but has added a fixed sample stage, just like a lab microscope would have, which holds the sample securely in front of the head. The optical head folds into the stage so that the package fits into a jacket pocket, and contains the camera and top and bottom illuminators, both of which are adjustable.
Although it’s still early days, the microscope has already been sold into micro-engineering applications such as electronics; microbiology research; universities and science centres, including the Eden Project; animal health; and for home use and education.
‘IoLight sees its role as developing the portable functionality, giving more people access to better microscopy in more places,’ Monk said. ‘We think that existing microscope manufacturers will continue to make beautifully engineered and expensive microscopes, which will have a place in the lab for the foreseeable future. However, high resolution portable microscopes will add another dimension of flexibility, optimising workflow and image sharing.
‘There will be applications that go portable very quickly and others that take longer to adapt. We are looking forward to learning what the market wants from a portable microscope.’