From wrinkle removal and plumping power, to instant hydration and antioxidant action, ever more cosmetics are flooding the market with claims of combatting the inevitable signs of ageing. Given this, cosmetic testing houses are turning to a range of optics-based methods to assess if ingredients and products are safe, and truly live up to the hype.
For Dr Nikita Radionov, commercial manager at Laboratoire Bio-EC, microscopy is a firm favourite. As he puts it: ‘Be it hydration studies, anti-ageing studies, many labs can perform clinical studies for cosmetic products but microscopic analysis produces very robust results and convincing evidence [of a product’s efficacy].’
During in vivo clinical studies, skin samples can be taken from volunteers using strips of tape that remove the three or four outermost stratum corneum layers of the skin. In a typical scenario, researchers may be testing the efficacy of a skin barrier repair product, where the replenishment of skin lipids can be deemed a desirable therapeutic target.
Here, immunostaining of samples for light microscopy can reveal structures in the samples such as filaggrin, a filament aggregating protein that binds to keratin fibres in epithelial skin cells, and ceramides, waxy lipids that link molecules and create a barrier to prevent permeability. Bio-EC researchers also work with Bordeaux Imaging Centre, using electron microscopy to analyse the skin samples and garner detail on the organisation of, say, ceramide, lipids and sebum in that sample.
‘The microscopy results we get are so visual and convincing, and tell a story,’ said Radionov. ‘For example with melanin analysis we can take an image of the corneal site [on an epidermal layer] and see the tiny black granules of melanin inside the cell.
‘We can also visualise all the damaged proteins on the skin surface – if a product protects against oxidative stress, we can use images of protein damage to compare the results before and after its use,’ he added.
But beyond the more traditional fluorescence and electron microscopy methods, researchers are developing techniques to study skin further. For example, researchers have been optically clearing small skin samples, using clarification methods such as 3Disco – 3D imaging of solvent-cleared organs – to render samples transparent. These samples can then be analysed using a range of light microscopy methods.
In a recent example, researchers Eric Fernandez and Sylvie Marull-Tufeu, from the Laboratoire Application Cutanée, Yves Rocher, worked with biopsy punches from human abdominal skin taken after plastic surgery. The samples were stained with the fluorescence dye Rhodamine B, and then cleared, with the transparent samples analysed using laser scanning confocal microscopy.
Using their technique, Fernandez and Marull-Tufeu visualised the micro-architecture of the epidermis at different skin depths – from the outermost stratum corneum layer to the deeper papillary dermis. And they also went on to reconstruct the entire epidermis in 3D, showing exactly how complex the skin is.
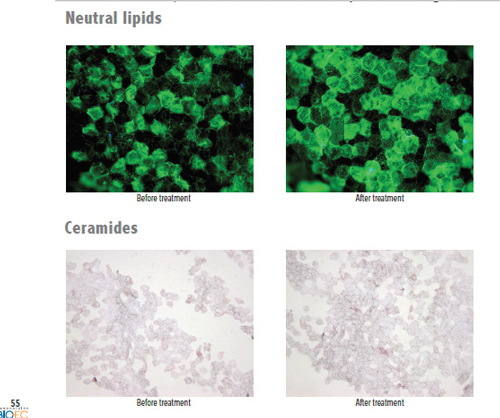
Image: Bio-EC
As Fernandez and Marull-Tufeu wrote in Skin Research and Technology: ‘This technique is inexpensive, fast and scalable. [It] could appear as a complementary tool for in vitro skin studies... to understand, compare or assess the effectiveness of a treatment with active ingredients.’
Radinov is excited by developments such as this. ‘At the moment the world is really creative and we are seeing more different approaches,’ he said.
Highlighting how such techniques could be used with different immunostaining methods to view, for example collagen and proteins, and also carried out on organs beyond the skin, he added: ‘When you see these beautiful images, they really look magnificent... it’s like the future is already here.’
Mapping moisture
Still, there’s more to cosmetic product testing than microscopy. Moisture mapping is widely used in clinical studies to assess the moisture content of the stratum corneum, as well as the micro-topography of the skin. The technique works on the same principle as the classic corneometer, which measures the dielectric constant of the skin, via capacitance sensors. The dielectric constant of skin changes with water content, so the tried-and-tested corneometer method precisely measures skin hydration.
However, moisture mapping uses capacitance imaging to generate maps of hydration distribution across the skin surface. Typically, a sensor containing tens of thousands of micro capacitors measures capacitance levels across the skin surface. These values are then transformed into a range of 256 grey levels to form a non-optical image.
Darker pixels represent the more hydrated, high capacitance regions, while clear pixels represent the dry spots of the skin, as well as any depressions or lines in the stratum corneum that impede contact between the sensor probe and skin.
‘With the classic corneometer you can see the immediate hydrating effect of a product.... but with our Moisture Map you get these images, as well as the values,’ said Radionov.
Interference fringe profilometry is another much-loved method used to assess skin topography in cosmetics testing. The technique calculates a phase image from an interference fringe image projection, providing data on the altitude of each point.
Widely used to evaluate the impact of anti-ageing products on wrinkles, especially crow’s feet, cosmetic testing companies are now looking for ways to use this method. As Dr Francis Vial, deputy CEO and scientific director of Spincontrol, highlighted, his company uses Aeva-HE fringe projection units, which also combine stereo imaging, to assess wrinkles on the neck, chest around the mouth and more.
Fringe projection units can provide different fields of view, depending on whether the user wants to study localised structures or larger regions of skin. ‘We will use a small field-of-view to study, say pores and fine lines at high resolution,’ said Vial. ‘We might look at the astringent effect on a pore in 3D – we can see if a product modifies the secretion of sebum and reduces the volume and diameter of a pore.’

Interference fringe profilometry is used to assess skin topography in cosmetics testing. Image: Spincontrol
The company will use a larger field of view to capture entire faces, as well as body parts such as the abdomen and thigh, with software creating 3D profiles of, perhaps, sagging at the jaw and the volume of the chin bone.
‘When we age, the adipose tissue in our cheeks decreases... and some cosmetic products use these molecules to have a plumping effect on the cheek. We can look at this,’ explained Vial. ‘[We also look at] any uplifting effects of the chest, and the effects of slimming products on the abdomen and volume of the thighs.’
In the early days of using interference fringe projection in cosmetic testing – more than 20 years ago – measurements would have to be acquired from silicone elastomer replicas of the skin surface. However, according to Vial, instruments are more accurate and less sensitive to motion, so readings can be taken directly from the volunteer. And thanks to developments in high-resolution digital light projectors and 3D measurement algorithms, imaging resolution has improved from around 200µm to nearly 1µm.

Interference fringe profilometry calculates a phase image from an interference fringe image projection. Image: Spincontrol
But beyond moisture mapping and fringe projection, Raman spectroscopy has become an indispensable testing method for clinical studies on volunteers. The technique uses near infrared light to examine skin in vivo, and according to Bio-EC’s Radionov, can provide reliable analysis to skin depths of around 30µm, meaning that stratum corneum effects can be investigated.
As part of its Raman-based testing, Bio-EC has been working closely with Professor Ali Tfayli from the University of Paris-Saclay. Tfayli has been developing Raman methods for many years, for example using Raman spectroscopy for in vivo imaging, to study lipid ageing within the stratum corneum. He has also used Raman confocal microscopy to study skin ageing.
Bio-EC has also worked with researchers from the University of Reims Champagne-Ardenne, using Raman spectroscopy and micro-imaging to monitor how different molecular weights of hyaluronic acids penetrate the skin. Topical delivery of molecules into the skin is a huge issue for cosmetics manufacturers, and research has confirmed that, in this case, Raman spectroscopy is an effective method to assess this.
‘With Raman, we can also evaluate hydration claims from product manufacturers – for the hydrating effect we can differentiate between two different forms of tyrosine [non-essential amino acid] and detect the more open form that is able to capture water,’ Radionov noted. ‘We can also analyse different natural moisturising factors, and the pools of protein and lipids. Raman is a very powerful technique but it is important to ask the right questions.’
Skin models
But testing isn’t all about in vivo methods. Ex vivo testing is essential to evaluate raw materials and finished products. Cosmetic testing companies have developed skin models that hold all the cells and characteristics of real human skin, and are designed to mimic the live response of in vivo human skin.
‘Several labs have declared they can perform studies based on living skin explants, but from what I can see, we are probably the only lab in the world making a business around this,’ said Radionov. ‘We carry out almost 360 studies a year with our main model, and we know how to keep the skin in a viable state for up to two weeks – this is very important.’
Ex vivo skin models, typically based on explants of human skin donated by volunteers, act as an intermediate stage between cell culture and true in vivo analysis. To cater for different tests, various models exist, containing different layers of the skin. For example, whole skin explant models include the epidermis, dermis and hypodermis layers, while other models may just include the hypodermis (adipose tissue). Crucially, these will be held in a culture medium to maintain the model’s viability.
‘We use a special culture medium, which we believe is unique,’ Radionov noted. ‘This keeps explants in a viable state and guarantees that whatever the donor, we can ensure the skin remains responsive to any external treatment or product applications.’
Bio-EC performs much of its usual test methods, including histology, transmission electron microscopy and Raman spectroscopy, on its skin models. As part of these tests, the models may be exposed to atmospheric pollutants and ultraviolet light to induce oxidative stress, inflammation or DNA damage. In addition, the skin may also be subjected to infrared light, bacteria, even small wounds, so that healing processes can be monitored.
However, as Radionov points out, the demand to assess the effects of blue light is rising. Some research has indicated that blue light, also known as high energy visible light and emitted from smartphones and other devices, can lead to hyperpigmentation and premature photoageing of the skin, and also disrupt circadian rhythms. ‘Blue light is very trendy right now... and cosmetic producers are beginning to think they can protect skin against blue light, as well as UVA and UVB light,’ he said.
To test the effects of pollutants and light on skin models, Bio-EC has developed various exposure chambers. For example, in the project Urbaskin, the company worked with researchers from French universities, as well as the Bio-Europe, Genex and Uriage Dermatological Laboratories to develop Pollubox, a transparent chamber with nebuliser system that sprays clouds of gases that may contain heavy metals or hydrocarbons, onto the skin model.
In a second project, custom-made chamber Solarbox has been built to expose skin models to different wavelengths of light and visible light. ‘This is a dark, non-transparent chamber with light emitters and filters so we can control the dose and density of the light,’ said Radionov. ‘Control is always important... we need to know which proteins are modulated by the stress and at what point in time.’
He added: ‘In another idea, we want to add more stress factors, such as pollution and UV light – this combination of different stresses allow us to get much closer to reality.’
Gtting closer to reality is what it’s all about. According to Radionov, research is now underway to also develop more sophisticated skin models that better mimic living skin. For example, skin model Perfex Vivo ‘reproduces’ blood flow via continually circulating culture medium throughout its layers. ‘This model is much closer to reality, and all our measured parameters correlate well with measurements from volunteers,’ said Radionov.
In another project, Bio-EC researchers are developing ‘nerve skin’, in which neural stem cells are added to the skin model, which then differentiate to neurons, so researchers can study reactions to stress and treatments. ‘This really is a very nice and beautiful concept,’ says Radionov. ‘And in each case, the idea is to create a skin model that is closer to the gold standard of clinical tests.’
Optical elements for aesthetic laser applications
Many dermatological and skin care procedures such as hair and tattoo removal, acne and scar reduction, and skin revitalisation are reliant on lasers for their respective treatments, where structures within the skin are modified using the photo-thermal and photo-acoustic effects produced by laser light energy.
Many aesthetic lasers used for dermatological treatments employ intra-cavity or extra-cavity optics or optical assemblies. Here, we examine a few cases where optical elements are utilised within these systems to couple, shape, or direct laser light for application to patients’ skin.
Optics in skin rejuvenation
Skin rejuvenation procedures often come in the form of ablative (skin tissue is vaporized) and non-ablative (skin remains intact) laser treatments. Some non-ablative laser treatment methods utilize non-uniform, fractionated beam delivery. Whilst non-fractionated lasers act on the entire projected surface area of the skin, fractionated lasers target multiple, equally distributed portions of the projected area.
Fractionated beams consist of several high-intensity zones surrounded by lower-intensity zones. The higher-intensity zones heat select portions of the target tissue to temperatures sufficient for a first treatment (e.g. collagen shrinkage), while the lower-intensity zones provide sufficient energy to the surrounding tissue for a second treatment. This process stimulates collagen regeneration to ‘fill in’ wrinkles and make skin look younger.
Fractionated laser beams are achieved by coupling the output beam from a laser source with an optical system that modifies the beam profile to a large-diameter beam with a non-uniform energy profile. Differing configurations of fibre bundles, axicon lenses, and diffractive or refractive lens arrays may be leveraged to provide non-uniform energy output beams. Figure 1 demonstrates how a diffractive element and lens could be combined to create an array of focal points.
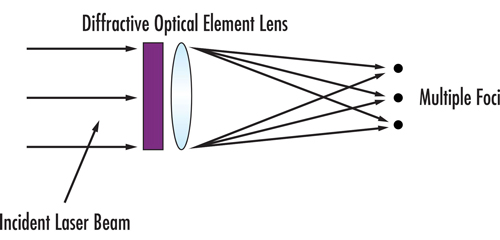
Figure 1: Conceptual generation of spots from a diffractive beamsplitter and lens
Optics in tattoo removal
Tattoos are permanent designs made of pigment that is injected into the skin’s dermis. The dense layer of pigment/ink that is created is too much for the body’s immune system to remove, and so it becomes entrenched below the dermis-epidermis interface. To remove tattoos, pulsed laser light is used to break down the larger ink particles into ones small enough to be engulfed by the immune systems’ phagocytes which then naturally remove these from the body as waste.
Laser tattoo removal procedures commonly employ solid-state lasers, e.g. Alexandrite, pumped by a second solid-state laser, e.g. Nd:YAG, with a q-switch and frequency converting elements, an example of which can be seen in Figure 2. In this case, the first laser cavity includes a highly reflective mirror and output coupler mirror. This output coupler is a partially reflective mirror, either flat or curved, which allows part of the circulating intracavity power to be released from the first laser cavity. The second laser cavity, in this case, would include a high reflecting pump mirror and an output coupler mirror since the second 1064nm solid-state laser is pumped by a portion of the initial 755nm output beam.
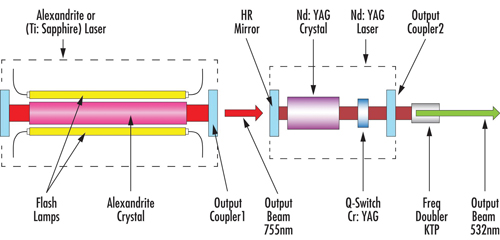
Figure 2: Typical layout of a solid-state laser pumped by a second solid-state laser with a q-switch and frequency converting elements, commonly used for tattoo removal
Aesthetic laser systems such as these vary by gain material, pulse intensity, and pulse duration depending on the application and precision needed for a given treatment. For example, due to the different optical absorption of the various colours of tattoo pigment, an Alexandrite laser can remove blue, purple, and green tattoos, while an Nd:YAG laser can remove black tattoos.
Laser light beam delivery
Typical primary or pump laser dermatology units consist of a main body inside of which the primary or pump laser is stored, an articulated mirror arm or optical fibre, and a handpiece at the end, usually with an optical assembly inside, which shapes the laser beam and directs it to the patient’s skin. Sometimes, these handpieces also include a second laser gain material which is pumped by the primary unit.
Coupling the output laser beam of the primary laser into the gain material of a second laser can be accomplished in a variety of ways. Figure 2 shows a direct approach. However, depending on pulse intensity, pulse duration, and wavelength, an optical fibre may be used to transmit the beam to a hand-held device.
Figure 3 shows a schematic drawing of a handpiece including a solid-state laser pumped via an optical fibre by another solid-state laser. The beam exiting the optical fibre is collimated by several lenses before being directed into the gain material. The output beam of the handpiece can further be optically modified by a beam shaper before it is used to treat the skin of a patient.
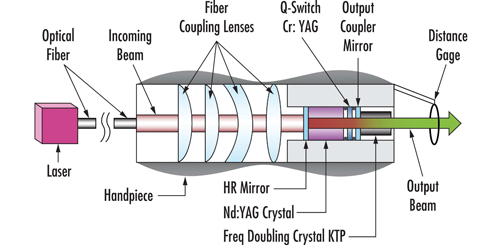
Figure 3: A schematic drawing of a handpiece including a solid-state laser pumped via an optical fibre by another solid-state laser
Edmund Optics proudly manufactures laser optics used in skin care.
Enquiries about complete systems should be directed to the skin care device manufacturers.