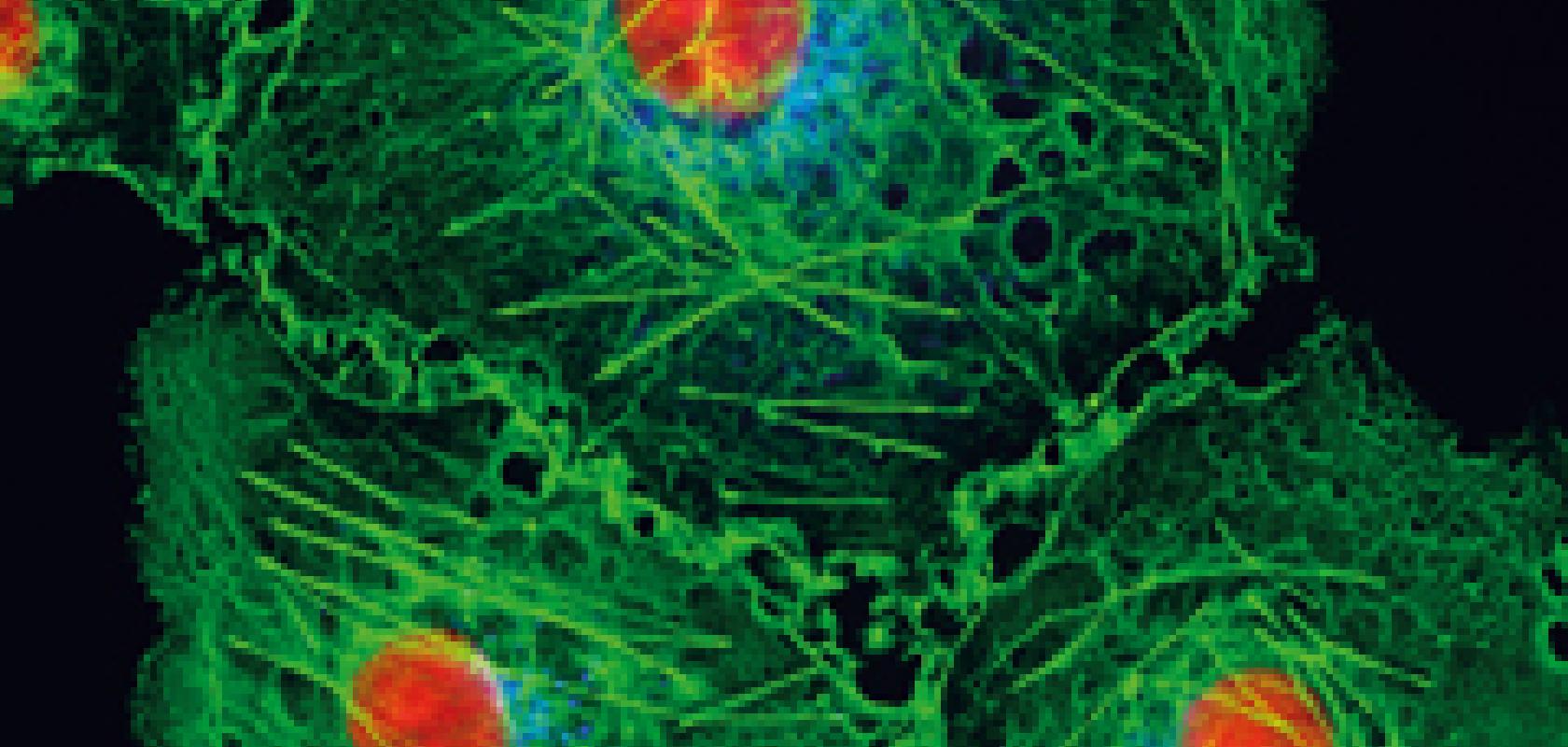
With approximately 86 billion neurons, the human brain is an incredibly complex organ and one that is still nowhere near to being fully understood. How light is used to study the brain has changed considerably over the last 10 or 20 years, progressing from straightforward imaging, to monitoring neural functions, and now there is a branch of neuroscience called optogenetics that uses light to excite or promote a function in a neuron.
‘Optogenetics is really a big change in the paradigm of looking at and understanding the way the brain works,’ commented Marco Arrigoni, a director of marketing at laser manufacturer Coherent. ‘I like to make the comparison to the subway map of London. Imaging is the equivalent of creating a map of all the lines. Optogenetics and signalling is the equivalent of having the subway map, plus controlling the trains and seeing where the trains go all of the time.’
The imaging aspects centre on confocal fluorescence microscopy, a technique that images dyes in the tissue as they fluoresce in response to light. An action potential, where neurons propagate a voltage signal from one to another to communicate, can be observed in this way. Dyes that fluoresce according to concentrations of calcium or other ions – the basis of the signal – can be used to monitor action potentials and indicate brain activity.
‘This is functional imaging,’ said Arrigoni. ‘You don’t just look at the shape of the cell or a neuron – which is morphology – but you can actually monitor the signal being transmitted from one neuron to another.’
Optogenetics takes this one step further by using light to excite or promote a function within a neuron, typically one that’s been genetically modified to express light-sensitive ion channels. Rather than just imaging action potentials, for example, light can be used to induce an action potential firing, or turn neurons on or off. This gives scientists a tool with which to gain greater insights into brain activity.
In order to control neural activity, neurons are first treated with light-sensitive proteins known as rhodopsins. Channelrhodopsin-2 (ChR2), for instance, responds to blue light, so a 460-470nm laser can activate the neurons treated with this protein. Alternatively, cellular expression can be turned off through the use of proteins known as halorhodopsins, which respond to light in the green part of the electromagnetic spectrum at around 550nm.
Traditional methods for functional analysis of neurons have relied on direct stimulation by tiny electrodes; however, scientists could not study specific regions of the brain in this way. The light-activated proteins allow scientists to turn neurons on or off selectively and precisely, opening up investigations into the structure and function of neural networks.
Dr Kavita Aswani, senior applications scientist, life sciences at photonics company Excelitas Technologies, commented that the beauty of optogenetics is its ability to study different physiologies within the brain by targeting specific populations of cells without affecting their neighbours.
Optogenetics also holds the potential for modulating activity of human brain circuits involved in neurological disorders such as Parkinson’s disease, or for restoring vision loss by helping reconnect neurons. ‘You can favour the so-called plasticity of neurons [with optogenetics] – how the neurons can reset connections after they have lost them,’ Arrigoni remarked. ‘If you think about the London Underground – if you have a train that is derailed, can you direct another train on a different line to get back to the station?’
There are two different ways in which optogenetics is typically performed. One is in a living animal, a mouse for instance, where a fibre probe would be attached to the brain. ‘You would have the other end of the fibre going off to a light source or to a camera. You turn on the light, induce the change, and then take a picture or record the event,’ Aswani explained.
For this type of setup, tiny fibres are required to deliver light into the brain of a small mammal such as a mouse. ‘We [Excelitas] are used to providing fibres that would go into a microscope, and they are quite large – between 1mm and 3mm. But for optogenetics, customers want 100µm or smaller, and in order to get light into a fibre that tiny there are huge challenges – just because of physics there is only so much light that you can squeeze into a tiny fibre,’ Aswani added.
The second method for performing optogenetics is on cultured cells or a tissue sample mounted on a microscope stage. ‘In that case, you would have the light coming through the microscope objective lens – you could use lasers, LEDs or even xenon lamps, because you’re relying on the microscope optics to focus the light onto the sample,’ Aswani said.
Femtosecond lasers
LEDs, continuous wave (CW) lasers or femtosecond lasers can be used in optogenetics experiments. LEDs will illuminate tens of thousands of neurons simultaneously, whereas a more localised population of neurons can be highlighted with a CW laser. Laser light can be focused on specific cells through the optics of a microscope.
In order to go deeper into the tissue sample or animal brain with greater precision, femtosecond lasers operating in the infrared are the best option. Femtosecond lasers are often integrated into two-photon microscopy systems, which use two photons of infrared light to excite the imaging probe.
Infrared light is scattered less than visible light in tissue meaning it can penetrate deeper. ‘In a typical sample, if you use a visible laser in a confocal microscopy setup you can go maybe 100-150µm deep under favourable conditions,’ Arrigoni noted. ‘If you use a femtosecond laser, you can typically go something like 800µm deep in the brain, and some “hero” results are about 1.5mm.
In addition, there is less photo-bleaching in the sample. This is partly because infrared light is less damaging to tissue, but also because of the nonlinear process involved when exciting with a femtosecond laser, explained Arrigoni. ‘If you imagine that you focus a visible laser on your sample, this light, this excitation, is throughout the entire sample,’ he said. ‘You can think of a cone of light that gets to a focus point – even if you only want to excite the tip of the cone, with visible light you end up exciting all of the tissue within that cone.
‘If you use a femtosecond laser, you are doing a nonlinear process, so you excite the sample only at the tip of the cone, and not above or below this tip,’ Arrigoni said. ‘If you excite less, you damage less.'
Although CW lasers are still the most popular choice, because, in part, they cost less – a CW laser might cost $5,000, whereas femtosecond lasers typically are priced between $150,000 and $250,000 – in the microscopy world, the market for confocal microscopes is well over a thousand per year, noted Arrigoni, with Coherent selling several hundred multiphoton systems per year.
Fibre lasers
It’s not only femtosecond lasers that have found their place within the neuroscience space; fibre lasers are also in demand for optogenetics research.
This is because, until two or three years ago, multiphoton systems were used mostly with titanium sapphire lasers, which are desirable because the wavelength can be tuned between around 680nm and 1,080nm.
The most common imaging probe for optogenetics has been green fluorescent protein (GFP); however, within the last five years, there has been an increased interest in red-shifted fluorescent imaging probes, or red fluorescent protein (RFP). GFP is activated at 920-950nm, whereas RFP responds to light shifted towards the infrared at between 1,050nm and 1,200nm. ‘This wavelength is really at the end of where titanium sapphire lasers work, so you cannot achieve enough power using one of these lasers,’ commented Arrigoni.
‘But fibre lasers are quite powerful at exactly 1,040-1,080nm, and they can produce many watts of power in this wavelength. So because of this interest in the RFP, there is really a specific place for these types of fibre lasers – they are being added more to optogenetics experiments,’ he added.
Smart money
The field of neuroscience has benefited from the launch of various funding programmes over the last couple of years. The most notable has been the BRAIN (Brain Research through Advancing Innovative Neurotechnologies) initiative in the United States that was set up in 2013. The programme has already invested $200 million into brain research, and in October it announced a further $100 million of funding for research institutions and universities carrying out neurological studies.
The University of California, Los Angeles (UCLA) was awarded $2.3 million in the new funding round. UCLA researchers aim to build a new generation of miniature fluorescent microscopes to image and manipulate the activity of large numbers of brain cells in mice that are moving freely in their natural environments. The tiny microscopes, which are expected to weigh less than 3g, will visualise individual neurons expressing calcium triggered fluorophores, which emit when specific wavelengths of light from the microscope are shone on them.
The researchers hope to adapt the mini-microscopes to allow scientists to control the activity of neurons remotely, and allow both monitoring and control with wireless technology.
‘Just like the Human Genome Project was for genomics, eventually the aim of optogenetics and the BRAIN project is to be able to treat neurological disorders ranging from depression to Alzheimer’s and Parkinson’s,’ said Aswani. ‘The BRAIN initiative helps to move the technology forward because there is more funding for researchers to perfect the technique and eventually test it on the human brain.’
Other funding programmes for brain research include the Human Brain Project (HBP) in Europe, Brain/MINDS in Japan, and a project which is soon to start in China. ‘The HBP is mostly a computational exercise in my opinion, using super computers to monitor the brain’s function. The BRAIN initiative is very technology-orientated, and light plays a big role in that,’ Arrigoni remarked. ‘It moves the focus to the optical study of the brain and its function on a neuronal stage.’
Optogenetics in humans
Although optogenetics is still in the research stage, the prospective ability to control signalling in the brain has a huge potential for treating human neurological disorders, from depression to Parkinson’s and dementia. So, what will it take to get to this stage, and is this even achievable?
‘As you can imagine, it will take a lot of research – getting these proteins into a human is a challenge, and there are all sorts of questions relating to how the proteins would affect the brain, and how you would deliver the light source to the brain,’ Aswani remarked.
‘You also have to bear in mind that the human brain is the most complex organ imaginable. We are starting with smaller organisms, such as the zebrafish and mice, whose brains are also complex but nowhere close to the human brain in terms of connectivity and complexity,’ Aswani continued. ‘Research is taking baby steps, but once we figure out how things work in the animals, then the experts will move onto humans.’
--
Removing brain tumours

Hyperspectral technology has been used to differentiate between healthy and cancerous brain tissues as part of the European project, HELICOID (Hyperspectral Imaging Cancer Detection), launched earlier this year.
The aim of the project is to improve the removal rate of tumours by identifying residual cancerous tissue during surgery. According to the project organisers, this will be particularly important in the treatment of brain tumours, where cancer cells are difficult to isolate with standard imaging techniques.
Headwall Photonics worked to develop the spectral imaging sensor as part of the Belgium-led HELICOID project. Early results obtained with the Headwall Hyperspec imaging system have been impressive.
One of the hallmarks of hyperspectral imaging is its ability to identify objects or disease conditions based on the chemical composition of tissue within the field of view of the sensor. By working closely with medical collaborators, the sensors were tuned to the precise spectral features of interest. By offering a precise definition of the boundaries of the cancer tissue in real-time, hyperspectral imaging can potentially accelerate cancer diagnosis and help surgeons remove a tumour while leaving healthy tissue untouched.
Edward Boyden at the Massachusetts Institute of Technology, and Karl Deisseroth at Stanford University and Howard Hughes Medical Institute, have both been awarded a Breakthrough Prize in life sciences for work on optogenetics. Each received $3 million, as did three other recipients of the life sciences prize.
The winners were honoured at a star-studded ceremony in Silicon Valley, hosted by Seth MacFarlane, with a live performance by Pharrell Williams, and Russell Crowe and Hilary Swank among those presenting the awards.
The Breakthrough Prize was founded by Sergey Brin and Anne Wojcicki, Jack Ma and Cathy Zhang, Yuri and Julia Milner, and Mark Zuckerberg and Priscilla Chan. The prize money totalled $21.9 million for awards in life sciences, fundamental physics and mathematics.
Optogenetics relies on light-sensitive proteins, originally isolated from bacteria and algae. About 10 years ago, Boyden and Deisseroth began engineering neurons to express these proteins, meaning the neurons could be stimulated or silenced selectively with pulses of light.
More recently, Boyden has developed additional proteins that are even more sensitive to light and can respond to different colours. Scientists are now using optogenetics to reveal the brain circuitry underlying normal neural function, as well as neurological disorders such as autism, obsessive-compulsive disorder, and depression.