Scientists at the Institute of Post-LED Photonics (pLED), at Tokushima University in Japan, have developed a new fluorescence microscopy method for studying dynamic biological processes.
The fluorescence lifetime microscopy technique uses frequency combs and no mechanical parts to observe dynamic biological phenomena. The work was published in Science Advances.
Conventional fluorescence microscopy only captures fluorescence intensity, which can vary depending on experimental conditions and the concentration of the fluorescent substance.
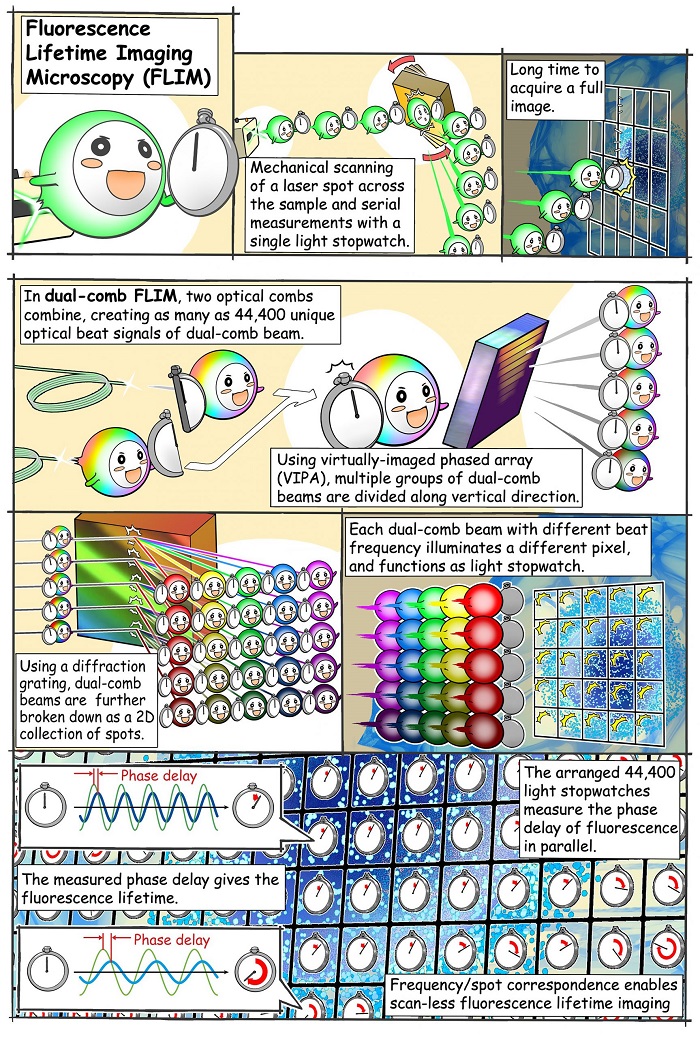
The new method measures both fluorescence intensity and lifetime. It does not require mechanical scanning of a focal point, but instead produces images from all points in the sample simultaneously, enabling a more quantitative study of dynamic biological and chemical processes.
Professor Takeshi Yasui, who led the study at pLED, commented: 'Our method can be interpreted as simultaneously mapping 44,400 light stopwatches over a 2D space to measure fluorescence lifetimes – all in a single shot and without scanning.'
Thanks to its speed and high spatial resolution, the scientists believe the microscopy method will make it easier to exploit the advantages of fluorescence lifetime measurements.
'Because our technique does not require scanning, a simultaneous measurement over the entire sample is guaranteed in each shot,' Yasui added. 'This will be helpful in life sciences where dynamic observations of living cells are needed.'
In addition to providing deeper insight into biological processes, this new approach could be used for simultaneous imaging of multiple samples for antigen testing, which is already being used for diagnosing Covid-19.
When a fluorescent substance is irradiated with a short burst of light, the resulting fluorescence does not disappear immediately but decays over time in a way that is specific to that substance. The fluorescence lifetime microscopy technique makes use of this phenomenon – which is independent of experimental conditions – to quantify fluorescent molecules and changes in their environment.
Fluorescence decay is extremely fast. To capture it, the scientists used an optical frequency comb as the excitation light for the sample. The fluorescence is focused onto a high-speed single-point photodetector, and the measured signal used to calculate the fluorescence lifetime.
Principle of operation. Manga courtesy of Suana Science YMY
An optical frequency comb is essentially a light signal composed of the sum of many discrete optical frequencies with a constant spacing in between them. The target sample is irradiated with a pair of excitation frequency comb signals, which decompose into individual optical beat signals (dual-comb optical beats) with different intensity-modulation frequencies, each carrying a single modulation frequency. The key here is that each light beam hits the sample on a spatially distinct location, creating a one-to-one correspondence between each point on the 2D surface of the sample (pixel) and each modulation frequency of the dual-comb optical beats.
Because of its fluorescence properties, the sample re-emits part of the captured radiation while still preserving the aforementioned frequency–position correspondence. The fluorescence signal captured by the photodetector is mathematically transformed into the frequency domain. The fluorescence lifetime at each 'pixel' is calculated from the relative phase delay that exists between the excitation signal at that modulation frequency versus the one measured.
Using optical frequency combs in microscopy techniques holds promise for developing novel therapeutic options for various diseases, the scientists say.


